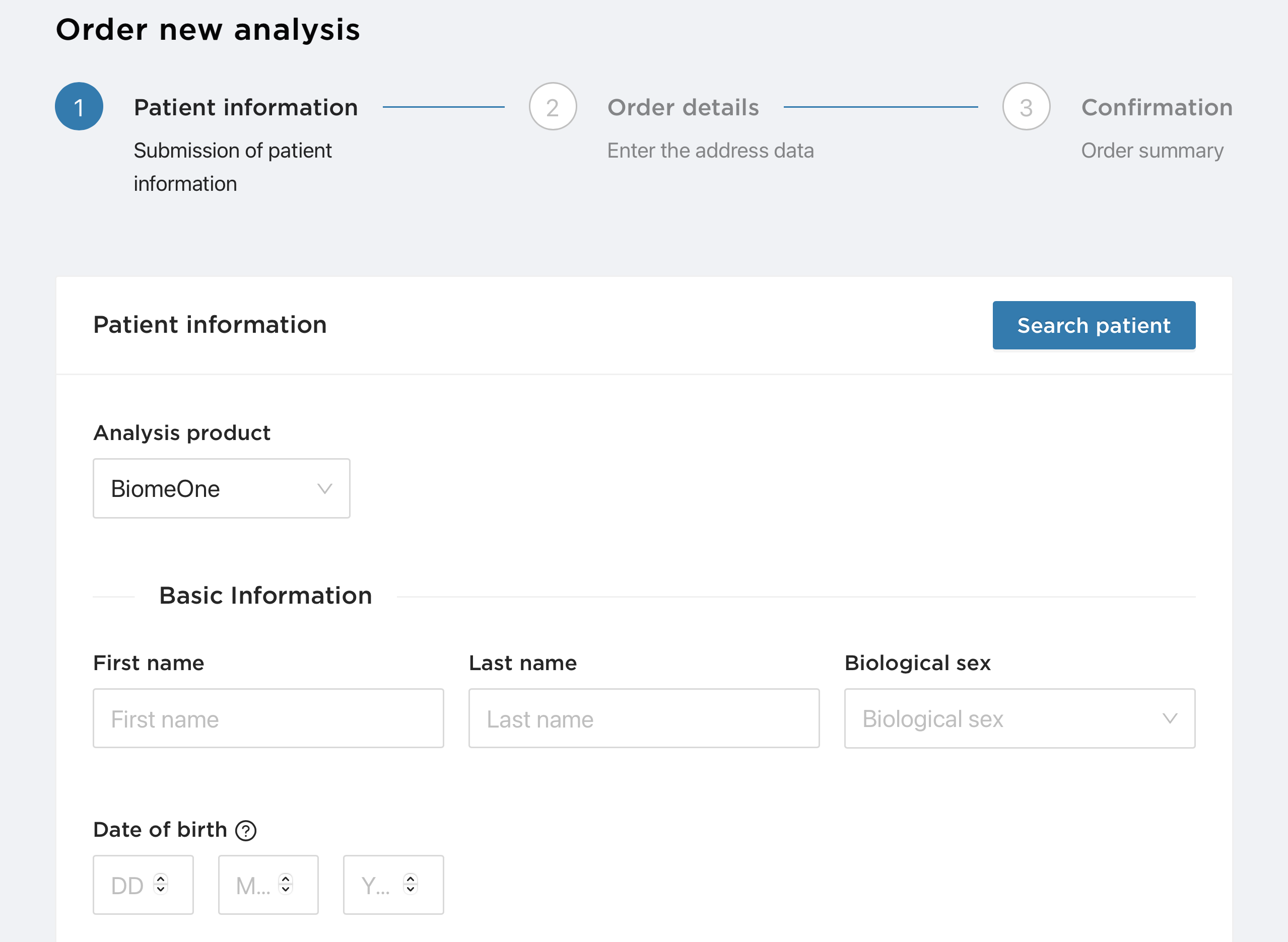
If you place an order for the BiomeOne Analysis test kit you will be asked to enter the patient information. If you are ordering a new analysis for a patient who has already been added to the patient list, you do not need to add the basic information section. This section will be filled in automatically as soon as you find the patient in your list using the search function.
Analysis test kit you will be asked to enter the patient information. If you are ordering a new analysis for a patient who has already been added to the patient list, you do not need to add the basic information section. This section will be filled in automatically as soon as you find the patient in your list using the search function.
You will need to provide the following personal information of the patient:
Patient name and last name
Biological sex
Date of Birth
BiomeOne Analysis is intended for biological men and women between the ages of 18 and 90 who have been diagnosed with stage III/IV non-small cell lung cancer, renal cell carcinoma or melanoma. More details about the general description of the product and its intended use can be found here.
Analysis is intended for biological men and women between the ages of 18 and 90 who have been diagnosed with stage III/IV non-small cell lung cancer, renal cell carcinoma or melanoma. More details about the general description of the product and its intended use can be found here.
You will be asked to provide clinical information about the patient. This section must be updated each time you order a new analysis, both for new and existing patients.
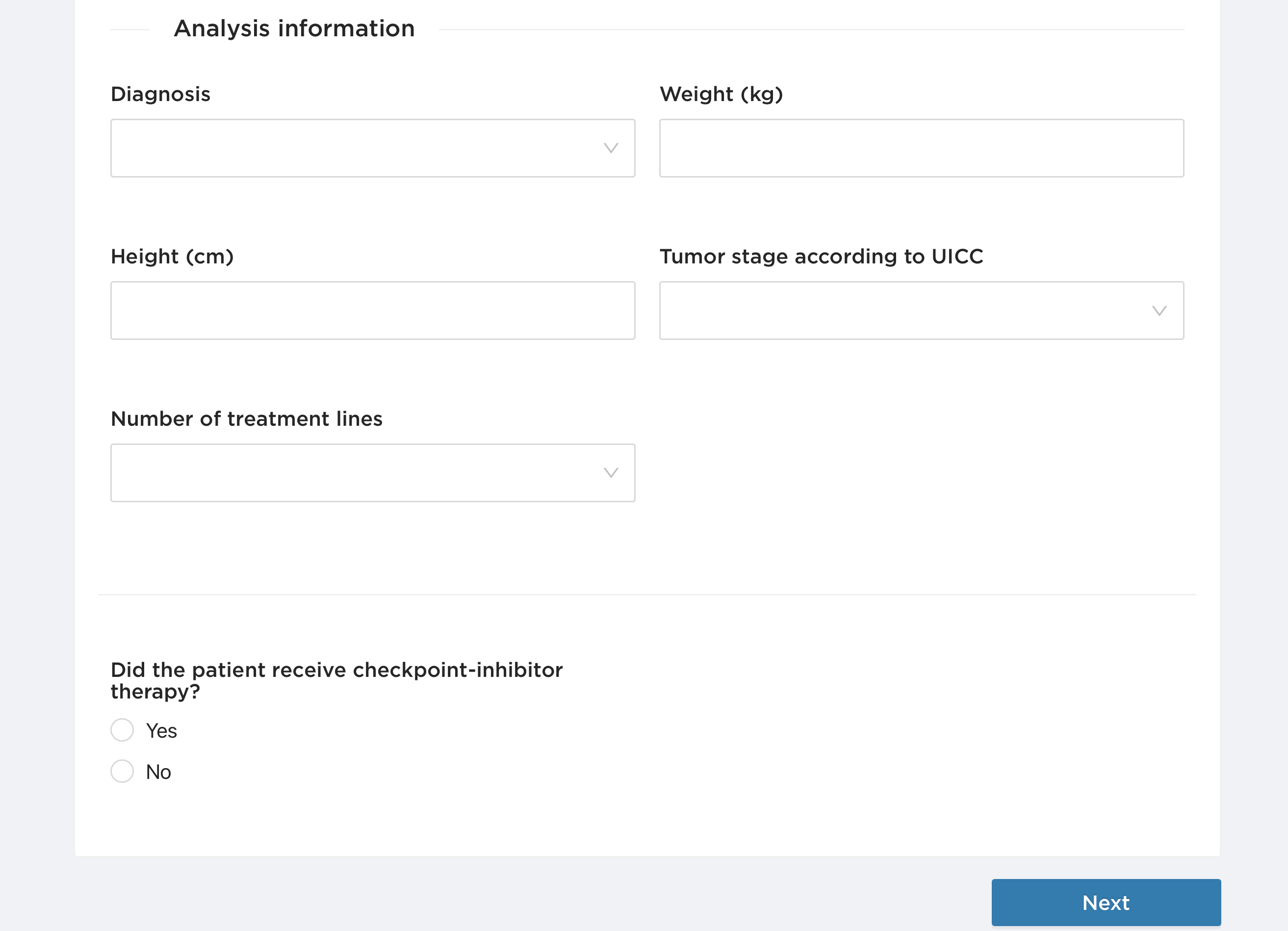
In the Diagnosis field, you can choose between melanoma, non-small cell lung cancer, renal cell carcinoma or other diagnoses.
You can also enter the patient's weight and height.
The tumor stage according to UICC allows you to classify the tumor stage into I, IIa-b, IIIa-c, IVa-b and unknown.
You can also provide information on the number of treatment lines (0-10).
Another relevant question is whether the patient has received checkpoint inhibitor therapy before the BiomeOne Analysis is performed.
Analysis is performed.
If you answer “yes” to the previous question, you will be asked about the type of immune checkpoint inhibitor administered to the patient (CTLA-4, PD-L1, PD-1), the duration of treatment in weeks, the type of clinical response of the patient to the immunotherapy administered (complete responder, partial responder, stable disease or progressive disease) and whether the patient had any immune-related adverse events. If the patient has experienced adverse effects, you will be asked to rate the severity on a scale from 0 (asymptomatic) to 5 (death).